Abstract
INTRODUCTION
Graft vs host disease (GVHD) is a rare complication after solid organ transplantation ( ̴ 1-2% with liver and ̴ 5.6% with intestines), but is associated with high mortality (70-80%). In contrast to GVHD following hemopoietic stem cell transplant, bone marrow infiltration by donor T lymphocytes leading to cytopenia ( ̴ 80%) is a common manifestation of GVHD after solid organ transplantation (Murali et. al., 100(12), Transplantation, January 2016). The risk factors associated with GVHD occurring after solid organ transplantation have not been well characterized, but single institution studies have suggested donor/recipient (D/R) HLA mismatch, ABO incompatibility and gender mismatch playing important roles. However, some studies have identified that similarities in HLA type which permit mismatches unrecognized by the recipient as a common mechanism preventing rejection of donor lymphocytes, which then can cause GVHD in the recipient. In order to evaluate potential risk factors, we reviewed the transplant data of patients in whom GVHD was listed as the cause of death from United Network of Organ transplant database (UNOS), the database that contains all US organ transplant data.
METHODS
From the UNOS database we obtained information of patients that underwent liver or intestinal transplantation between 1987-2020 in the US. The patients for whom GVHD was reported as the cause of death were identified. Baseline D/R and transplant variables were collected. Patient or transplant related characteristics were presented via descriptive statistics. Corresponding P-values were determined using Chi-square test for categorical variables and two-sample t-test for continuous variables. The incidence of mortality caused by GVHD was estimated using the cumulative incidence method, accounting for non-GVHD related death as a competing risk. All statistical tests were two sided, and a P-value < 0.05 was considered significant.
RESULTS
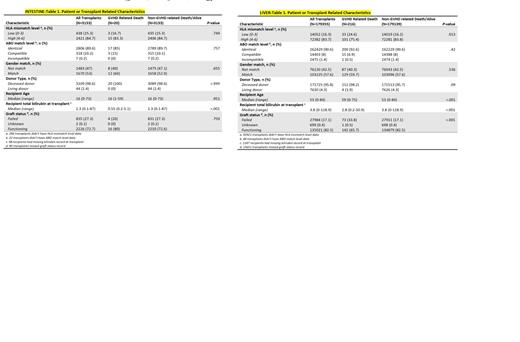
Of a total of 179,355 patients that underwent liver transplantation, 216 (0.1%) patients had GVHD identified as the primary cause of death (GVHD group). The HLA mismatch information was available in 86,434 patients (48.2%). HLA mismatch was grouped into low level (0-3) and high level (4-6). Low level HLA mismatch was 24.6% in the GVHD group compared to 16.2% in patients that are alive or died of non-GVHD related causes (non-GVHD group). High level mismatch was 75.4% in GVHD group and 83.8% in the non-GVHD group (P=0.013). Other risk factors including gender mismatch (40.3% vs 42.5%, P=0.536), ABO incompatibility between D/R (0.5% vs 1.4%, P=0.42) and use of live donors (1.9% vs 4.3%, p= 0.09) were similar between the two groups. Patients in the GVHD group were older with median age of recipient being 59 years compared to 53 years in the non-GVHD (P<0.001). Graft failure was more common in the GVHD group compared to non-GVHD group (33.8% vs 17.1%, P<0.001).
Of a total of 3,153 patients that underwent intestinal transplantation, 20 (0.6%) patients had GVHD identified as the primary cause of death (GVHD group). The HLA mismatch information was available in 2859 patients (90.6%).The high-level HLA mismatch (83.3% vs 84.7%, P=0.749), gender mismatch (40% vs 47.1%, P=0.655) and ABO incompatibility (0% vs 0.2%, P=0.757) between D/R were similar between the GVHD group and non-GVHD group among intestinal transplant patients.
Conclusions
In patients that undergo hematopoietic stem cell transplant, HLA and gender mismatch between D/R have been recognized as risk factors for GVHD. Based on the largest analysis of solid organ transplant database, traditionally considered GVHD risk factors like HLA and gender mismatch between D/R do not appear to be significantly associated with severe GVHD leading to death. Recipient age and graft failure are significantly associated with GVHD related deaths in liver transplant patients. These findings suggest that other risk factors for severe GVHD leading to death after solid organ transplant than those previously reported in single institution studies should be examined and underscore the need for additional studies.
Grunwald: Med Learning Group: Other; Agios: Consultancy; Astellas: Consultancy; Daiichi Sankyo: Consultancy; Janssen: Research Funding; Amgen: Consultancy; Pfizer: Consultancy; PRIME: Other; PER: Other; Karius: Consultancy; Sierra Oncology: Consultancy; Blueprint Medicines: Consultancy; Incyte: Consultancy, Research Funding; AbbVie: Consultancy; Bristol Myers Squibb: Consultancy; Stemline: Consultancy; Gilead: Consultancy; Cardinal Health: Consultancy; Trovagene: Consultancy; MDEdge: Other. Copelan: Amgen: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal